This article covers a LOT of information. Don’t use this to avoid doing your own research – use it to guide your research. And, especially with the newer medications, new data is coming out all the time. By no means is this information static – new information is coming out all the time and once you’ve downloaded this (to your computer or your brain), I can’t get in there and update it!
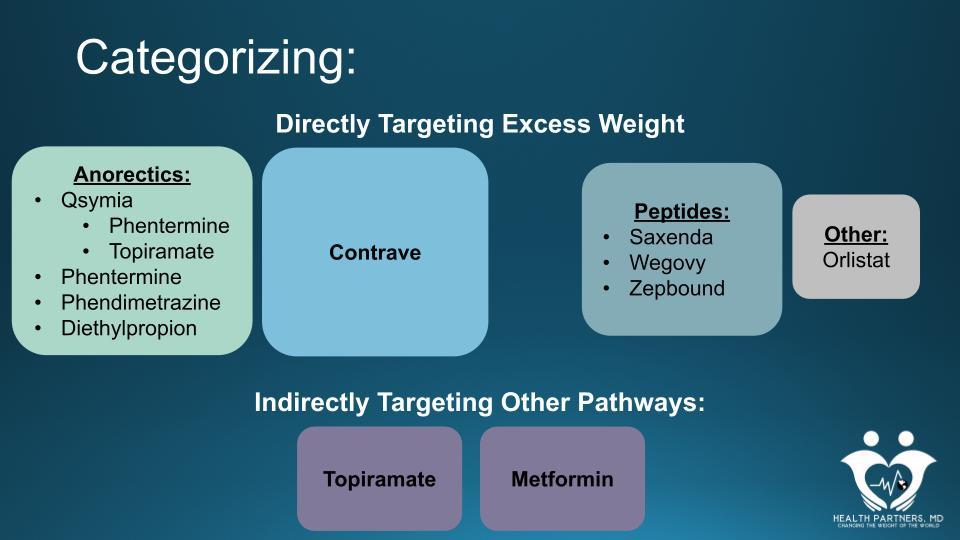
The following picture helps explain the way the following information is organized and presented. I know it looks like a PowerPoint slide – it comes from a slide deck I use to take clinicians a bit deeper into how to practice comprehensive Obesity Medicine. If that’s ever something you are interested in (or know someone who is), check it out on this website. I love sharing what I know!
Anorectic Medications:
- Phentermine (trade names: Lomaira®, Adipex-P®)
- Phendimetrazine (trade names: Bontril PDM®, Bontril Slow Release®)
- Diethylpropion (trade names: Tenuate®, Tenuate Dospan®)
Anorectic medications live in the category of sympathomimetic amines, or stimulants. There’s a lot of misinformation about these medications and I often encounter a lot of fear from healthcare providers and pharmacists when discussing them. Yes, these medications are classified as stimulants, but they are very similar to Ritalin and Adderall – and we give these to small kids every day without worrying about it. Yes, they have side effects – and yes, they have contraindications – but they are much less scary than most people think! For those of you who take care of women who might become pregnant during treatment (and who don’t have any contraindications), these are good drugs to consider. The half-life is very short (less than a day) so stopping them at the first sign of pregnancy means very little exposure – similar to stimulants used to treat ADD/ADHD.
Contraindications:
- known allergy or hypersensitivity
- pregnancy
- nursing
- history of CVD (CAD, stroke, certain arrhythmias, CHF, uncontrolled HTN)
- hyperthyroidism
- narrow-angle glaucoma
- agitated states
- history of drug abuse
Warnings/Precautions:
- avoid concomitant use with other stimulants
- elevations in heart rate and/or blood pressure due to anorectic medications are typically very minimal, however, blood pressure monitoring in anyone with HTN is recommended
Phentermine/Topiramate ER: (Trade Name: Qsymia®)
If you aren’t familiar with this drug, you should be. It’s a great little drug and quite a few people are hyper-responders to it. Most people tolerate it pretty well too – and it’s dirt cheap compared to GLP-1s. There are more contraindications to it – mostly because it’s a combination of two drugs. We’ve already reviewed the contraindications to phentermine – the same ones hold true here, as well, but we also have to add in the contraindications to topiramate. I’m sure you are familiar with this topiramate. It’s been around for a long time. It doesn’t have the best reputation in terms of side effects, but the good news is that the dose of topiramate in this drug is low compared to the doses used to treat migraine headaches – and it’s delivered in an extended-release format, so you don’t get big peak concentrations – which helps a lot. For those of you who manage women who might become pregnant – please pay close attention to the precautions about this drug listed below. Topiramate is a big no-no in pregnancy!
Contraindications:
- known allergy or hypersensitivity
- pregnancy
- nursing, history of CVD (CAD, stroke, certain arrhythmias, CHF, uncontrolled HTN)
- hyperthyroidism
- narrow-angle glaucoma
- agitated states
- history of drug abuse
- nephrolithiasis
- concomitant MAOI use within 14 days
Warnings/Precautions:
- pregnancy testing is recommended before initiation and monthly during therapy. Women who can become pregnant should be advised of the potential risk to a fetus and to use effective contraception during therapy – take this seriously – as a previous OB/GYN, I beg you!
- titrate QOD dosing for at least 7 days if discontinuing the med from 15/92 mg dose to reduce seizure risk
- avoid concomitant use with other stimulants
- elevations in heart rate and/or blood pressure due to anorectic medications are typically very minimal, however, blood pressure monitoring in anyone with HTN is recommended
- may potentiate hypokalemia caused by non-potassium-sparing diuretics
- may cause (reversible) paresthesias
- may potentiate CNS depressants such as alcohol
- use caution in people with renal impairment
Naltrexone/Bupropion XR (Trade Name: Contrave®)
This is another combination drug. It’s also a great little drug for some people. Although there are fewer hyper- responders to this drug, when it works for someone, it really works for them.It also has the added bonus of not being a) a controlled substance and b) prohibitively expensive. If a woman becomes pregnant while taking this drug, it’s also nothing to panic about. It’s not technically contraindicated in pregnancy (meaning we don’t have data to indicate it’s unsafe) – although deliberate weight loss is contraindicated in pregnancy, so stopping the medication is advised. Anorexia nervosa or bulimia is officially listed as a contraindication on this medication only, although it applies to all AOMs.
Contraindications:
- known allergy or hypersensitivity
- uncontrolled HTN
- seizure disorder
- narrow-angle glaucoma
- bipolar mood disorder
- anorexia nervosa or bulimia
- undergoing abrupt discontinuation of alcohol, benzodiazepines, barbiturates, and/or antiepileptic drugs
- use of other products containing bupropion
- chronic opioid use
- during or within 14 days of taking monoamine oxidase inhibitors
Warnings/Precautions:
- pregnancy was removed as a contraindication in 2020, however, the prescribing information recommends discontinuing when pregnancy is recognized
- can cause false positive urine test results for amphetamines
- tamoxifen and digoxin levels may be decreased
- use caution in people with renal impairment
Peptides/GLP-1 Agonists:
- Liraglutide (trade names: Saxenda®, Victoza®)
- Semaglutide (trade names: Wegovy®, Ozempic®)
- Tirzepatide (trade names: Zepbound®, Mounjaro®)
Because these are the medications you are likely using the most, I’m going to do a bit of a deeper dive here compared to the other medications. There are a few things to be aware of. These medications have been on the market for some time but weren’t widely used until recently. We are still learning about them.
Tirzepatide has a warning that oral contraceptive agents may be less efficacious for a month after initiation of the medication as well as for a month after any dose increase. This isn’t noted for liraglutide or semaglutide, but it doesn’t hurt to be cautious. I’m not sure they studied this.
In rats and mice, these drugs cause dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in both genders. It is unknown whether this extends to humans. Since many practitioners are approaching obesity treatment with the philosophy that AOMs are required indefinitely, I imagine we will find out eventually if this applies to humans. I don’t want my patients to be a part of that experiment and, as I’m sure you have gathered, I don’t view these medications as being required life-long in every patient. Regardless, we are told to counsel patients regarding the potential risk of medullary thyroid carcinoma and inform them of symptoms of thyroid tumors.
There is a warning with all of them about an increased risk of pancreatitis. I don’t start these medications on anyone with a history of pancreatitis. It seems too risky. I’ve had patients die from pancreatitis.
Hypoglycemia can occur when used with insulin and or an insulin secretagogue such as sulfonylureas. If you start someone on one of these medications, please make sure they are well aware of the risks. Ideally, titrate down their insulin and/or other anti-diabetes medications before letting them develop hypoglycemia.
Because nausea can be significant with this class of medications, they can precipitate vomiting and/or decreased fluid intake. There are quite a few case reports of patients presenting to the ED with severe dehydration. In some of these cases, renal impairment has been reported, which has sometimes required hemodialysis. With proper counseling and supervision, this should be avoidable, but you have to talk to patients about it.
Monitor for depression or suicidal thoughts and discontinue if symptoms develop. These medications affect the reward system of the brain (as do other AOMs). When reward is removed, it can affect mood and pleasure and, in susceptible individuals, may have downstream consequences. We are just starting to understand this and how to monitor it and address it, but it’s good to be aware of it.
Finally, there was recently a study demonstrating that patients taking these medications have an increased risk of developing nonarteritic anterior ischemic optic neuropathy (NAION). Although this is associated with type 2 diabetes, at least in the preliminary report, the increased risk extended to those patients without a diagnosis of diabetes. The warning has not reached the drug package insert, but it’s worth watching.
Contraindications:
- known allergy or hypersensitivity
- pregnancy
- nursing
- personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2
Warnings/Precautions:
- acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed. Discontinue promptly if pancreatitis is suspected; do not restart if pancreatitis is confirmed
- causes dose-dependent and treatment-duration-dependent thyroid C-cell tumors at clinically relevant exposures in rats and mice
- monitor heart rate to evaluate for possible heart rate increase
- renal impairment has been reported post-marketing, usually in association with dehydration,
- monitor for depression or suicidal thoughts and discontinue if symptoms develop
- consider reducing the dose of concomitantly administered insulin secretagogues or insulin to reduce the risk of hypoglycemia
- delaying gastric emptying has the potential to impact the absorption of concomitantly administered oral medications. Monitor patients on oral medications dependent on threshold concentrations for efficacy and those with a narrow therapeutic index (e.g., warfarin) when concomitantly administered
- advise patients using oral hormonal contraceptives to switch to a non-oral contraceptive method or add a barrier method of contraception for 4 weeks after initiation and for 4 weeks after each dose escalation. Hormonal contraceptives that are not administered orally should not be affected (** tirzepatide only)
- be cautious in people with gastroparesis
Orlistat (Trade Name: Xenical®)
I will briefly mention this medication because it’s officially classified as an AOM, but to be completely honest, I’ve never once prescribed this medication – despite practicing Obesity Medicine for over ten years. One, it’s avaialble over the counter at a significantly lower price and two, it’s not a great medication.
Contraindications:
- known allergy or hypersensitivity
- pregnancy
- nursing
- chronic malabsorption syndrome
- Cholestasis
Warnings/Precautions:
- gastrointestinal events may increase when orlistat is taken with a diet high in fat
- patients should be strongly encouraged to take a multivitamin supplement that contains fat-soluble vitamins. The supplement should be taken at least 2 hours before or after the administration of orlistat
- may increase the risk of cholelithiasis
- may increase the risk of urinary oxalate and kidney stones
- rare post-marketing reports of severe liver injury and pancreatitis have been reported

